Companies that count on BSI Life Sciences
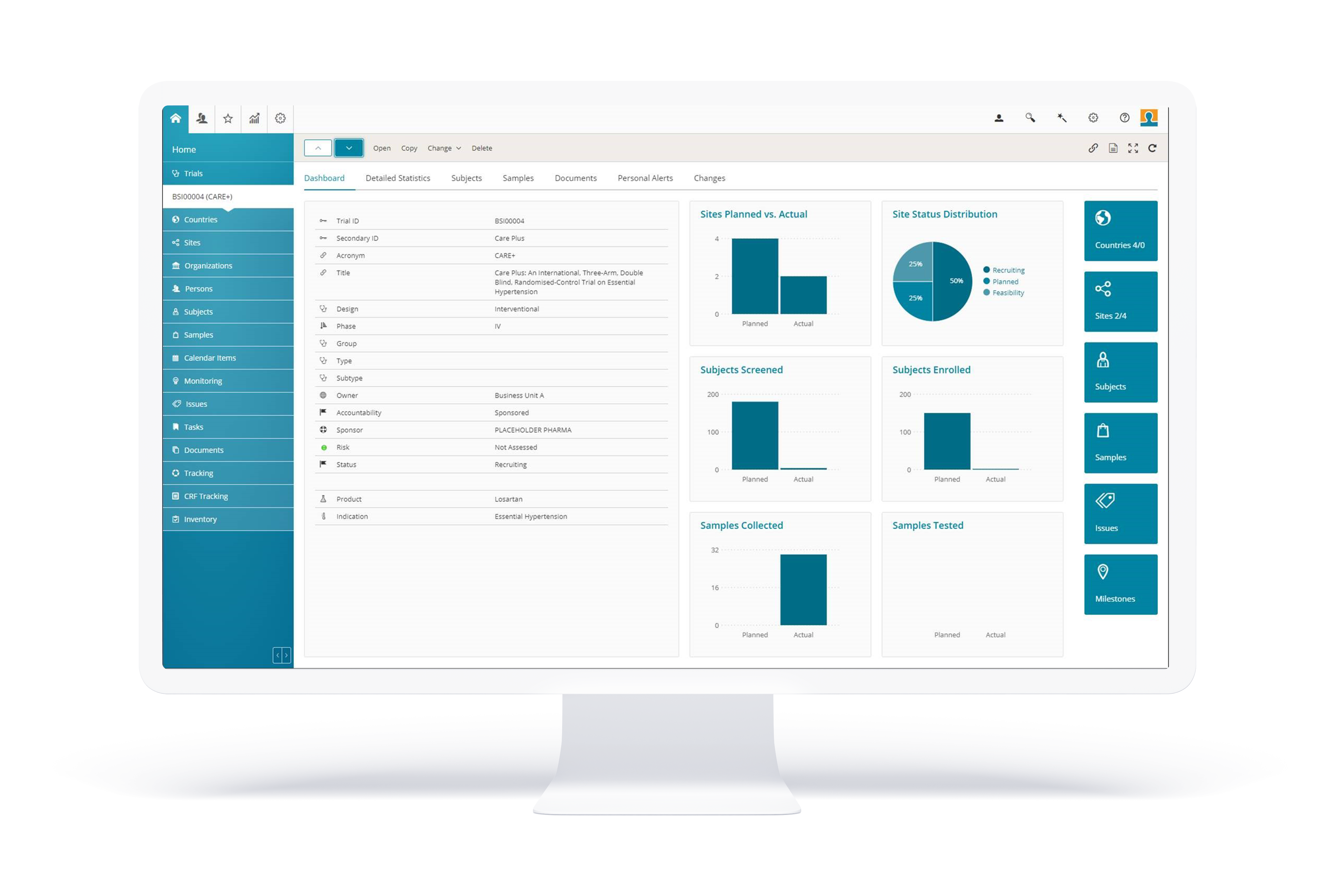
BSI CTMS
The most efficient tool for your clinical trials
BSI CTMS is an innovative software solution for all phases of clinical trials. It includes CTMS (Clinical Trial Management Software), eTMF (electronic Trial Master File), Study Startup, LMS (Learning Management System), and TSM (Trial Supply Management).
Many benefits
- Features smart data visualization;
- Simplifies the management of partner and sponsor access rights;
- Facilitates compliance with legal data protection requirements;
- Integrates easily with your existing systems;
- Comprehensive reporting capabilities.
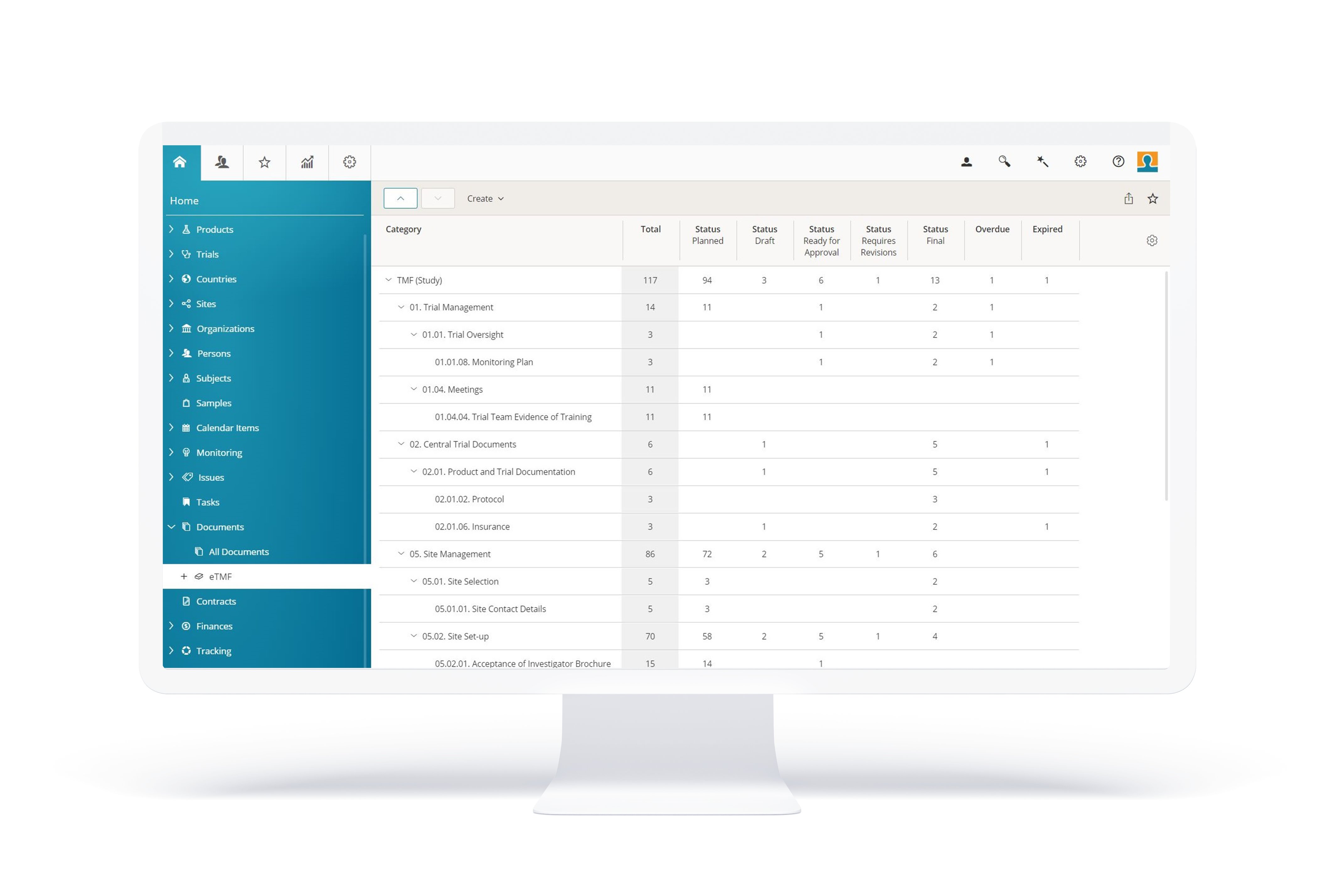
BSI eTMF
The feature-filled eTMF solution for your clinical trials
BSI eTMF is a user-friendly software solution for managing your Trial Master File and offers an excellent price-performance ratio.
Many benefits
- Very easy to use;
- Simplifies the exchange of documents with study centers (sites);
- Facilitates compliance with legal data protection requirements;
- Available as a stand-alone solution or as a component of BSI CTMS.
Increased productivity
for all stakeholders
Regardless of the area in which you conduct clinical trials, the solutions from BSI Life Sciences provide everything you need.
About BSI
Would you like to know more about BSI?
We make life sciences software for people and have been developing software for more than 25 years. What makes our software solutions, made in Switzerland, Germany and the United States, stand out is their innovative and user-friendly design. We focus our resources on staying ahead of the curve and delivering practical and cost-effective clinical trial software, thus helping our customers continually optimize their clinical development processes. And that's what we love to do.
BSI Life Sciences is a unit of BSI Business Systems Integration AG, one of Switzerland's most successful and experienced privately held software companies. BSI has provided solutions for life sciences since it was founded in 1996 and has become a market leader in CRM and marketing automation solutions for insurance, banking and other industries in Germany and Switzerland. BSI – that is 500+ employees, 9 offices in Switzerland, Germany and the United States, and more than 25 years of market success – all driven by the genuine values we live by every day: We love software. And we love our customers.